John Zone, MD
Dr Zone, an expert in systemic disease and Co-Director of the Immunopathology Lab at the University of Utah, discusses the medications that we use to treat these various conditions. Most of the therapies discussed in this presentation are off-label unless otherwise noted.
What does all of this cost? The table below depicts the numbers from the University of Utah pharmacy.
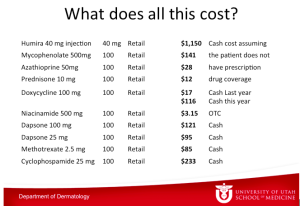
Dr Zone has said this before, if he gets pemphigus, he wants to take rituximab. He is convinced that rituximab is not only the best treatment, but it is the cheapest in the long run.
IgE
IgE is more under the purview of the Allergist, so it isn’t discussed much in dermatology. It is a monomer and is present in very low concentrations in the body. Its synthesis is 25 to 2000-fold less than other immunoglobulins making it difficult for us to measure. It lasts in the blood for 48 to 72 hours so it has a very short half-life. IgE doesn’t activate C’ and instead binds on mast cells for weeks to months. If you develop a sensitivity, once the IgE molecules bind to mast cells, they are going to be there for awhile no matter what you do.
The approach that we take to treat conditions like urticaria always astounds Dr Zone. We know that IgE binds to mast cells in our skin. When the mast cell becomes degranulated, it releases histamine, TNF, proteases, and heparin—the things that cause people to get symptoms of itching and swelling manifested as urticaria. We treat people with antihistamines– we give people something to block the effect of the histamine after it’s released. There are a number of papers studying the positive effects of TNF inhibitors in urticaria. At Dr Zone’s institution, they have treated about thirty people with fairly good results. If you have intractable urticaria, you are open to a lot of things. Over a longer period of time, there is release of prostaglandins and leukotrienes. There are prostaglandin inhibitors and montelukast that can be used as well for urticaria. Over the long run, we then see eosinophil recruitment and release of other cytokines.
Given this information on pathogenesis of urticaria, it is clearly better to block the effect of IgE and prevent the end results?
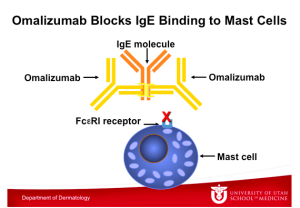
This graphic illustrates Omalizumab binding to an IgE molecule, which also takes place at the constant region of the IgE molecule. Note that IgE binds either to the FceRI receptor on the mast cell or to Omalizumab, but it cannot bind to both at the same time. Omalizumab inhibits the binding of IgE to the high-affinity IgE receptor FceRI on the surface of mast cells and basophils. Reduction in surface-bound IgE on FceRI-bearing cells limits the degree of release of mediators of the allergic response. Treatment with omalizumab also reduces the number of FceRI receptors on basophils in atopic patients.
Omalizumab is approved for asthma at this time and has recently been approved for refractory urticarial. To read more about omalizumab, the following references may be helpful:
- Pemphigoid
- G Ital Dermatol Venereol. 2012 Jun;147(3):251-7.
- Urticaria
- Curr Opin Allergy Clin Immunol. 2012 Aug;12(4):406-11
- Atopic dermatitis
- Clin Exp Dermatol. 2012 Oct 22
There are going to be a lot more studies on urticaria and atopic dermatitis with omalizumab. Dr Zone uses Omalizumab 150-375mg subcutaneously every two to four weeks. It costs anywhere from $500 to $2000 per month and he has been able to get insurance companies to cover it. Regarding side effects, anaphylaxis has been reported; however, omalizumab is rather well tolerated.
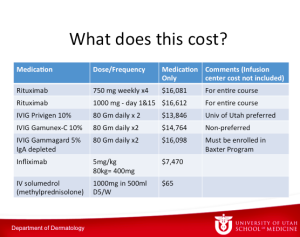
Intraveneous Immune Globulin in Autoimmune Diseases
IVIG is currently used for the following dermatologic disorders:
- Pemphigus vulgaris
- Pemphigus foliaceus
- Bullous pemphigoid
- Mucous membrane pemphigoid
- Epidermolysis bullosa acquisita
- Toxic epidermal necrolysis
- Necrotizing fasciitis
There is mixed information suggesting that IVIG works; but because these diseases are very severe, there is likely to be approval.
IVIG comes from thousands of donors and there is no data to show that one brand is better than another. Somewhere around one percent of the population are IgA deficient. If you are IgA deficient, you can have an anaphylactic reaction when IgA-containing immunoglobin is administered. Dr Zone believes that everyone should have a total serum IgA done before receiving IVIG. If you cannot do this, then the patient should be given the IgA depleted therapy. IVIG is used in smaller doses for immunodeficiency.
The therapeutic effects of IVIG most likely reflect the function of natural antibodies in maintaining immune homeostasis in healthy people. We think that antibodies are just for killing bacteria and viruses, but it’s not just that. Antibodies regulate our immune system. When you get someone else’s IVIG, you have a great chance of changing the regulation of your immune system.
The debate on toxic epidermal necrolysis (TEN) and IVIG continues. Ten or so years ago, Dr Zone was convinced that IVIG was the best treatment for TEN; but now he’s not totally sure. Huang and colleagues conducted a systematic review and meta-analysis looking at the efficacy of IVIG on the treatment of TEN. Seventeen studies were included. (Huang, et al. BJD 2012:167:424-432) The studies reported that pediatric patients had lower mortality than adults and adults with high dose IVIG had lower mortality compared to low dose IVIG (18.9% vs 50%). However, logistic regression failed to show a significant correlation with mortality.
What would Dr Zone do if he had TEN? After studying the papers, he would take IVIG.
IVIG for SJS/TEN Overlap and TEN:
- Start IVIG 1 gram per kg for 3 days or 0.75 gram per kg for 4 days
- If there is any concern for IgA deficiency, start IgA depleted IVIG until IgA levels are confirmed
- In cases of renal failure, obtain renal consult and consider 0.5 gram per kg for 4 days vs. short course, high dose steroids
Dr Zone believes that the survival is better if it is done carefully. He would also most likely take IVIG with progressive Stevens Johnson’s Syndrome. IVIG has been used for over thirty years for patients with primary or secondary antibody deficiency and has not been found to have long-term side effects. IVIG has been tested by PCR for hepatitis B and C and HIV. There have been no cases of HIV transmission and no recent cases of hepatitis transmission. Side effects of IVIG include:
- Headache, migraine
- Fatigue
- Anaphylaxis
- Aseptic meningitis
- Congestive heart failure
- Pulmonary edema
- Acute renal failure
- Leukopenia
- Hemolytic anemia
- Hepatitis
- Venous thrombosis
There are a group of people who believe that cyclosporine works for SJS/TEN. A study of 29 patients was published in the British Journal of Dermatology suggesting a possible usefulness of cyclosporine in SJS and TEN that needs to be confirmed. (Valeyrie-Allanore L, et al. Br J Dermatol. 2010;163(4):847-853.)
Takeaway point—If Dr Zone had SJS/TEN, he would take IVIG. If you didn’t want to give IVIG, cyclosporine is an option. Otherwise, you are in for a disease with a thirty percent mortality rate.
Scleromyxedma
This is a very rare disease and as dermatologists, we will likely see one case in our lifetime. A case series from Johns Hopkins reported eight of out ten patients with either 100 percent complete or partial response rate with IVIG treatment 2G/kg/month in divided doses. In the old days, there was no good treatment for scleromyxedma. Once again, if Dr Zone had scleromyxedema, there is no doubt that he would take IVIG. You do end up taking it indefinitely, but we try to decrease the dose and frequency.
Oral Corticosteroids
Dr Zone feels that he sees a lot more side effects from oral corticosteroids than immunosuppressive therapy. Before starting a patient on oral corticosteroids, it is important that you check their blood pressure as well as glucose, electrolytes and creatinine. Dr Zone gives H2 blockers or proton pump inhibitors to most people who are on therapy for more than two or three weeks. Gastritis and upper GI bleeding is a common side effect. For more than two or three weeks, Dr Zone also uses osteoporosis prevention. It is also important to monitor side effects every two weeks.
How can we prevent osteoporosis?
- Suggest Calcium carbonate plus D for short term (twice daily)
- Bisphosphanates
- Alendronate 5 mg daily or 35 mg. weekly
- Beware of esophagitis
- Calcitriol (Vitamin D3)
- .5-1.0 micrograms daily
- Beware of serum calcium
- Estrogen or Testosterone
You can start prednisone at approximately 1mg/kg/day. The patient can take the entire dose in the morning or BID early in the day. Alternating day therapy prevents adrenal suppression, but not osteoporosis. It is extremely important that you try to control the disease then minimize the dose, not the reverse. The reason that you want to taper steroids is to avoid rebound in the short term and manage adrenal suppression in the long term. If you want to evaluate for adrenal suppression, you can do an 8am cortisol and ask the patient not to take their steroid.
Dapsone
Dr Zone uses a lot of dapsone in his practice because he treats a lot of patients with dermatitis herpetiformis (DH). Dapsone inhibits neutrophil chemotaxis and attachment. It has neither an effect on antibody or complement deposition nor an effect on the reaction to gluten in DH. Dapsone simply blocks the inflammation in the skin.
Every patient on dapsone gets hemolytic anemia. Dapsone is a sulfone, meaning it has a double-bond oxygen—an incredible oxidant stress. What does that do to red cells? Red cells older than 90 days don’t have much G-6-PD because it decreases as cells age. They don’t have the ability to withstand oxidant stress. When you give dapsone, you’re going to hemolyze cells that are over 90 days old. If the red cell lifespan is 120 days and you give dapsone, you might hemolyze one quarter of the red cell mass. What does this mean? If you’re a young person you might feel a little fatigued; however, if you’re 80 and have congestive heart failure (CHF), it might end up exacerbating the CHF. But remember that everyone is going to get hemolysis. What happens? Your bone marrow starts to produce reticulocytes so all of a sudden the mean cell volume (MCV) goes up. When you treat someone with dapsone, you will see macrocytosis (because of the big reticulocytes) and a slight fall in the hematocrit, or you may see a fairly big fall in the hematocrit. With time, most people will compensate enough so that they’ll have enough reticulocytosis that they’ll raise their hematocrits back to the normal level. They’ll still be constantly breaking apart the red cells that are more than 90 days old because they can’t withstand oxidant stress.
Before giving dapsone, you should perform a baseline CDC and chem profile as well as a G-6-PD in Asians, African Americans, or those of southern Mediterranean descent. Start dapsone at 25mg daily and increase by 25mg weekly until the symptoms are under control. A CBC should be done weekly for the first four weeks, then monthly for six months, then semiannually. A chem profile should be performed at six months and then annually.
Dr Zone has treated somewhere around 1000 people with dapsone. He’s had two people who have developed leukopenia within two weeks. You can get aplastic anemia that will occur within the first four to six weeks. This is why you want to check the blood counts frequently in the beginning.
Mycophenolate mofetil (MMF)
MMF was approved by the FDA in 1995 for the prophylaxis of acute rejection in renal transplant patients. It is an ester of mycophenolic acid (MPA) and is synthesized to increase the bioavailability of MPA. It works by inhibiting leukocyte recruitment and glycosylation of lymphocyte glycoproteins involved in endothelial and intercellular adhesion and lymphocyte trafficking.
When you think about what we do as dermatologists whether it’s eczema or severe dermatitis, we are treating lymphocytes. MMF is a fairly good to drug to use in these cases. It’s effective as a corticosteroid-sparing agent in corticosteroid responsive dermatoses. MMF takes six to eight weeks to have an effect. You should start patients at 500mg bid. The most effective dose is 1000mg bid and the maximum dose is 1500mg bid. You may want to monitor levels if you are concerned about toxicity.
Dr Zone has seen the following side effects in patients receiving more than 2 g/d:
- GI disturbances (nausea, diarrhea, dyspepsia, abdominal pain)
- Persistent cough
- Reversible hematologic side effects
It is unknown as to whether or not MMF increases the risk of lymphoma and carcinogenicity. There is an increased risk of viral and bacterial infections.
Azathioprine
Dr Zone uses a lot of azathioprine and feels that it’s a great drug. Everyone should get a thiopurine methytransferase (TPMT) level before starting azathioprine as well as a CBC and comprehensive metabolic panel. Azathioprine is converted to 6 mercaptopurine within one hour. It works by inhibiting T lymphocyte function and Ig synthesis.
Dr Zone starts at 50mg/day and increases to 3mg/kg/day over one to two months. Some people get severe nausea and/or an acute febrile reaction. With severe nausea, most patients just can’t take the medication. With mild nausea, you can divvy up the dose to three times per day. It is important to monitor CBC for leukopenia and perform liver function tests for obstructive patterns and hepatocellular damage. You can taper steroids and continue the azathioprine.
Cyclosporine
Here’s the trick with this…the pharmacy may call you and ask if you want to use “modified” or “non-modified.” “Modified” cyclosporine (gengraf and neoral) has more consistent absorption than non-modified (sandimmune). They are NOT bioequivalent. It is important that you use one and stick with it. Before starting cyclosporine, you should perform a baseline CBC, metabolic profile and check magnesium.
Start cyclosporine at one to two mg/kg/day. Remember that hypertension and elevation of creatinine are limiting factors. Leukopenia may occur as well as hypomagnesemia. You should monitor CBC and metabolic profile monthly. Patients can also monitor their blood pressure at home.
Cyclophosphamide
This is used for ocular cicatricial pemphigoid and refractory bullous diseases as well as refractory vasculitis. If you haven’t used a lot of this drug, you should probably refer the patient to someone with experience. Cyclophosphamide can cause leukopenia as well as alopecia and gonadal damage. There is also a risk of leukemia and bladder cancer.