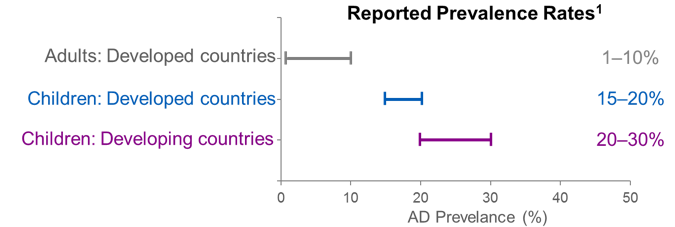
A real-world study evaluating adequacy of existing systemic treatments for patients with moderate-to-severe atopic dermatitis (AD-QUEST): baseline treatment patterns and unmet needs assessment
Presenters: Wei W1, Ghorayeb W2, Andria ML3, Walker V4, Chao J3, Schnitzer J2, Kennedy M3, Chen Z3; Belland A4, White J4, Silverberg JI5
Affiliations: 1Formerly of Sanofi, Bridgewater, NJ; 2Sanofi, Bridgewater, NJ; 3Regeneron Pharmaceuticals, Inc., Tarrytown, NY; 4Optum, Eden Prairie, MN; 5Northwestern University Feinberg School of Medicine, Chicago, IL
Objective: To evaluate potential unmet needs in the treatment of moderate-to-severe atopic dermatitis (AD) from a patient perspective.
Methods: Adults with an AD diagnosis in the last five years and a prescription for systemic treatment in the last six months were identified using claims data from Optum Research Database. Patients with self-assessed moderate-to-severe AD were invited to participate in monthly surveys over 12 months about disease signs and symptoms, quality of life (QoL) and AD treatment. We report baseline survey data.
Results: Of 6,000 patients invited to participate, 1,485 responded and 801 were eligible for inclusion (mean age: 45 years; female: 72%; Caucasian: 84%; employed: 79%; AD severity [Rajka & Langeland criteria]: moderate 74%, severe 26%). In the 12 months before baseline, 38 percent and 36 percent reported no remission or less than three months remission from AD. In the month before baseline, most reported using topical corticosteroids (64%) or calcineurin inhibitors (8%), some reported using antihistamines (38%), and a few reported using systemic steroids (11%) or immunosuppressants (5%). In the same period over 81 percent reported AD flares (1: 23%; 2: 20%; ?3: 38%), of which, 65 percent and 22 percent had partial or no recovery. Patients experiencing a flare versus those with no flare at baseline reported significantly worse POEM (13.5 vs. 6.2), peak pruritus NRS (worst itch in previous 24 hours: 6.3 vs. 3.5) and DLQI scores (8.6 vs. 3.7), and greater work productivity loss in previous seven days (8.2 vs. 3.4) (all P<0.001).
Conclusion: This suggests that despite standard-of-care treatments, adults with moderate-to-severe AD report disease symptoms, recurrent flares, and impaired QoL, suggesting unmet therapeutic needs.
Funding/Disclosures: Wenhui Wei is a former employee and current stockholder of Sanofi and an employee of Regeneron Pharmaceuticals, Inc. Eric Ghorayeb and James Schnitzer are employees of and stockholders in Sanofi. Michael Andria, Jingdong Chao, Martha Kennedy and Zhen Chen are employees of and stockholders in Regeneron Pharmaceuticals, Inc. Valery Walker, Angela Belland, and John White are employees of Optum, a company that received research funding for the current study. Jonathan Silverberg is a member of an institution that received research funding for the current study. This study was funded by Sanofi and Regeneron Pharmaceuticals, Inc.